
Ginger Vieira has lived with type 1 diabetes since 1999 along with a few other chronic illnesses! She is the Cofounder of the Diabetes Nerd Network and the Diabetes Nerd Marketing agency. Ginger has authored hundreds of articles and several books, including Exercise with Type 1 Diabetes, Pregnancy with Type 1 Diabetes, Dealing with Diabetes Burnout, and Stop Overeating During Lows. Over her 20-year career (so far) in diabetes, she’s produced hundreds of articles and videos about life with diabetes. Her background includes a Bachelor of Science in professional writing and certifications in coaching, personal training, powerlifting, and Ashtanga yoga. Find Ginger’s YOUTUBE, PODCAST, INSTAGRAM, and LINKEDIN.

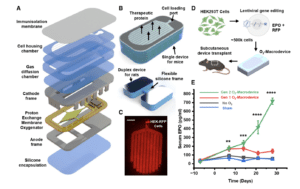
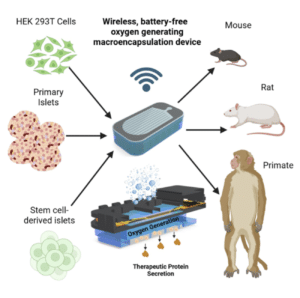 As exciting as this is, it’s important to keep our feet on the ground here.
As exciting as this is, it’s important to keep our feet on the ground here.